Stable One-Shot COVID Vaccine Deemed Safe and Effective
Paul Christian Gordon/Zuma
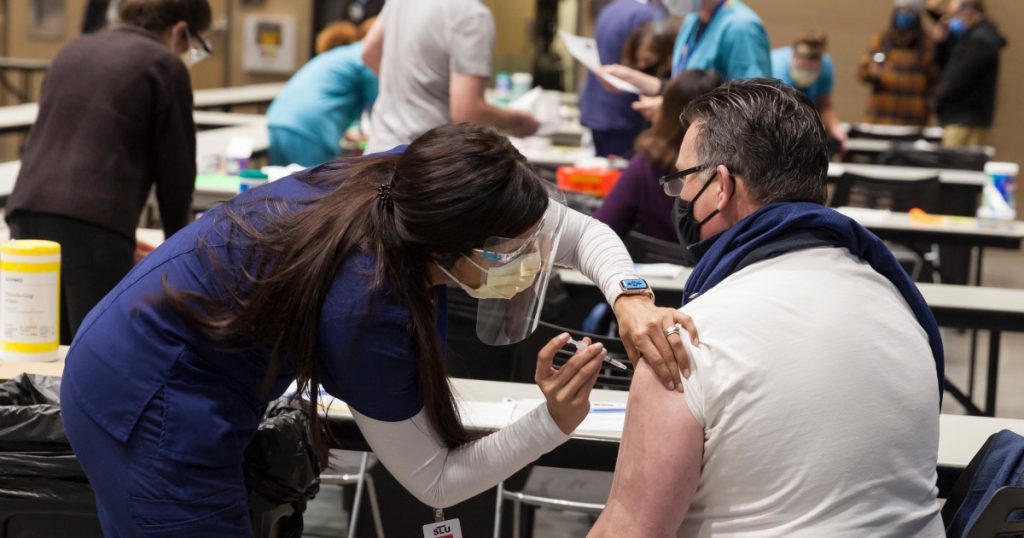
Let our journalists help you make sense of the noise: Subscribe to the Mother Jones Daily newsletter and get a recap of news that matters.Johnson & Johnson’s one-shot coronavirus vaccine is highly effective at preventing severe disease and death from COVID-19, the Food and Drug Administration found in analyses published today.
An international study found the vaccine’s efficacy rate to be 72 percent in the United States and 64 percent in South Africa, where a more contagious variant is spreading. While these figures are lower than Pfizer’s and Moderna’s efficacy rates, the vaccine greatly reduces the risk of hospitalization and death from COVID-19 and could play a key role in keeping the pandemic at bay. In a clinical trial of 40,000 people, no one who took the vaccine was hospitalized or died of COVID-19 after the vaccine took effect.
Plus, the vaccine requires only one shot and does not need special refrigeration—good news at a time when demand for vaccines greatly outstrips supply.
The FDA could authorize the vaccine for emergency use as early as Saturday, with 20 million doses available by the end of March and a total of 100 million doses delivered by the end of June.